Temporal seizure propagation patterns in epilepsy
A Bayesian model offers a promising tool to pinpoint seizure focus, potentially improving outcomes for drug-resistant epilepsy surgery.

1. Introduction
8. Conclusion
Introduction1
Managing focal epilepsy, therapy-resistant epilepsy and refractory epilepsy cases? New data supports personalized models for improved treatment and outcomes.
This approach offers potential for more targeted interventions and improved patient outcomes in managing convulsions.
A personalized approach to epilepsy surgery1
Imagine a future with more precise and effective treatment of focal seizures, offering a higher chance of seizure freedom. This research brings us closer to that reality.
Harnessing the power of individual patient data – seizure patterns, brain anatomy, and clinical expertise – this Bayesian model creates a personalized "virtual brain" alowing clinicians to visualize seizures spread in focal epilepy.
What does this mean for daily practice in managing focal seizures?
-

Refined surgical planning1
Pinpoint focal seizure origins and map their propagation pathways accurately, minimizing damage to healthy brain tissue and maximizing the chances of a successful outcome in focal epilepsy.
-

Personalized treatment strategies1
Identify patients benefiting from surgery and guide optimal approaches. Explore alternative treatment options, such as targeted drug therapies or neuromodulation techniques for refractory epilepsy.
-

Unlocking new therapies1
Deeper understanding of seizure dynamics opens the door to new interventions for focal epilepsy.
What is focal treatment-resistant epilepsy?1
Refractory or therapy-resistant epilepsy, observed in nearly 25% of epilepsy patients, is a class of epilepsy where medication fails to control seizures.
Focal epileptic seizures are characterized by seizures originating in one or more regions, generally referred to as an epileptogenic zone (EZ).
These focal seizures then propagate to other regions that are connected to areas in the EZ, generally referred to as a propagation zone (PZ).
Treatment of focal seizures1
Clinical treatment options in therapy-resistant epilepsy usually involves surgical resection of brain regions that are considered to be originating in the RZ.1
With success rates of epilepsy surgery, ranging between 50% and 60%, the need to develop better methods to identify EZs is evident.1
A Bayesian framework to identify epileptic seizure patterns1
A Bayesian framework based on a dynamical model of epileptic seizures, namely Epileptor, has been proposed by Vattikonda et al (2021) as a model to identify spatiotemporal seizure propagation patterns.1
In the field of neuroscience, Bayesian inference has been extensively and successfully used for model inversion by a class of models called dynamic causal modeling (DCM). DCM is a Bayesian framework to infer physiological mechanisms that can generate observations obtained from various neuroimaging techniques such as functional magnetic resonance imaging (MRI) and electroencephalography (EEG).
A Bayesian inference paradigm was used by the authors to perform model inversion. Bayesian inference offers a flexible framework to incorporate any prior knowledge such as plausible range of model parameters, dynamics of unobserved brain states, and prior hypothesis on the seizure focus.
By incorporating this information the parameter space is constrained. This allows for efficient exploration of posterior distribution of parameters.
Harnessing the power of individual patient data, this Bayesian model allows clinicians to visualize seizures spread in focal epilepsy. To improve management of drug-resistant epilepsy, this framework integrates patient-specific data (Fig. 1).1
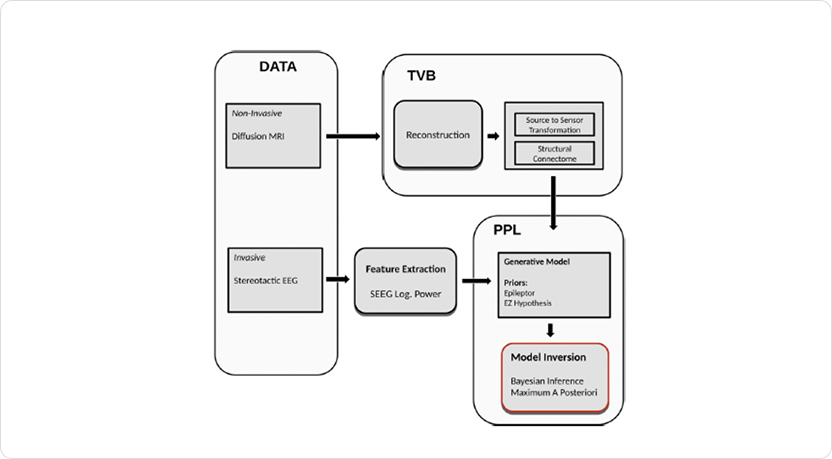
Fig. 1: Workflow for identifying seizure propagation patterns in focal drug-resistant epilepsy using a Bayesian framework. The process integrates patient-specific MRI and SEEG data to estimate structural connectivity and seizure dynamics, enabling personalized identification of the epileptogenic zone (EZ) and propagation zone (PZ) for improved surgical planning and treatment strategies. The figure is adapted from Vattikonda et al 2021 figure 1.1
-
The authors tested the model against a retrospective patient cohort of 25 therapy-resistant epilepsy patients who underwent surgery.
The cohort was divided into two groups based on the outcome the of surgery:
(1) Engel score I and II: patients who are either seizure-free or show rare disabling seizures
(2) Engel score III and IV: patients with minimal or no worthwhile improvement
Out of the five brain regions in the clinical EZ hypothesis, the model predicted four regions to be recruited by the seizure with one subcortical region (right hippocampus) in EZ and three regions (right thalamus proper, right amygdala, and ctx-rh-G-oc-temp-med-Parahip) as part of PZ.
In the patients who were seizure-free after treatment of focal seizures, model predictions showed a good match with the clinical hypothesis. In patients where surgery failed to achieve seizure freedom, the model predictions showed a strong mismatch.
Workflow for identifying epileptic seizure propagation patterns1
The process for identifying seizure propagation patterns involves the following steps:
(a) Estimating structural connectome (SC) and transforming from source to sensor space using diffusion, MRI data and electrode implantation, respectively.
(b) Extracting data features, such as log power over a sliding window, from the observed SEEG data.
(c) Defining a generative model that describes the joint probability density over the observations and Epileptor model parameters.
(d) Performing model inversion using MAP techniques to infer the model parameters and latent source states that best fit to the observed SEEG log power.
(e) Computing the seizure-onset times of all regions involved in the seizure by thresholding the inferred latent states.
(f) Identifying the EZ and PZ: regions with onset times within the lowest onset time and a small onset tolerance window are classified as part of EZ, while regions with onset times greater than this window are classified as part of PZ.
To test the model's accuracy, it was first validated against synthetic data. The Bayesian model accurately predicts seizure onset times in synthetic data (Fig. 2), demonstrating its potential for improved identification of EZ and PZ.1
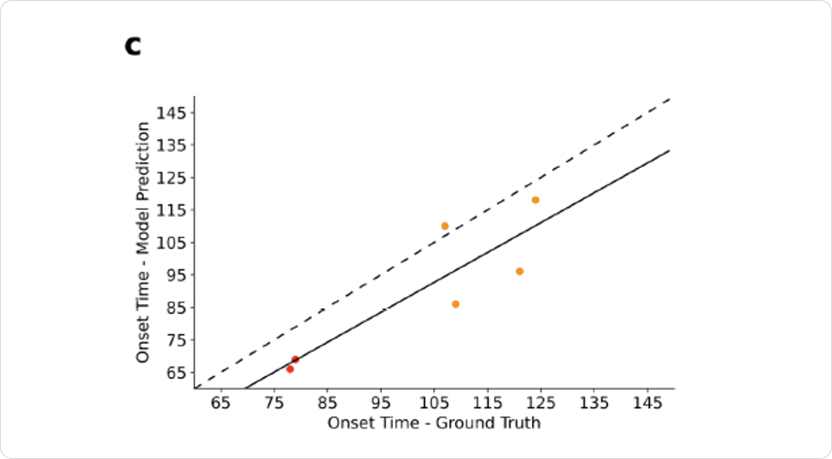
Fig. 2: The Bayesian model accurately predicts seizure onset times in synthetic data, demonstrating its potential for improved identification of the epileptogenic zone (EZ, red) and propagation zone (PZ, orange) in patients with epilepsy. The figure is adapted from Vattikonda et al 2021 figure 2c.1
Conclusion1
Applying a Bayesian framework to model epileptic seizure propagation offers a promising way to improve treatment of focal seizures. By combining prior knowledge with intracranial recordings, this research shows the potential of whole-brain models to simulate epileptic seizure dynamics. While further research is needed, this probabilistic model represents a significant step forward in the fight against treatment-resistant epilepsy, offering a powerful tool to improve outcomes and personalize treatment for focal epilepsy.
- Vattikonda AN, Hashemi M, Sip V, et al. Identifying spatio-temporal seizure propagation patterns in epilepsy using Bayesian inference. Commun Biol. 2021;4(1):1244.
 HarmoniaMentis Sverige
HarmoniaMentis Sverige